You may be asking yourself the question as to why a government panel would reject a prostate screening test that could save a life. This news is getting posted on many medical websites.
The panel decided that the harm of the screening test outweighs the benefit for preventing prostate cancer.
The following is an article from a Boston publication.
The US Preventive Services Task Force determined that based on evidence
from two large randomized trials, the lifesaving benefits of screening
were “at best very small” and were offset by overdiagnosis and
overtreatment of non-lethal cancers.
.“Our most optimistic estimate is that 1 out of 1,000 men screened will
avoid dying from prostate cancer” because of early detection via the PSA
test, said Dr. Michael LeFevre, co-vice chair of the task force. “We’re
not saying it’s zero. We’re leaving the window open for at least a
small benefit.”
“It’s hard to understand where they’re coming from,” said Dr. Anthony
D’Amico, chief of genitourinary radiation oncology at Brigham and
Women’s Hospital, in an interview.
D’Amico argued that the task force relied too heavily on data from a
flawed study and failed to consider making separate recommendations for
men in high-risk groups, such as those with a family history of prostate
cancer and African Americans, who have a two to three times greater
risk of dying of the cancer than white men.
The task force, which is comprised of 16 primary care physicians and
public health experts with no financial interests in tests or
treatments, issues screening and other preventive health recommendations
that tend to be more conservative than those of medical societies --
composed mostly of specialists who treat diseases detected through
screening -- or patient advocacy groups.
Former New England Patriots player Mike Haynes, a paid spokesperson for
the urological association, said in an interview that he was diagnosed
with prostate cancer in 2008, at age 55, after getting a free screening
PSA test at an NFL event for retired players, sponsored by the
urological association. He said he wasn’t told about any of the risks of
the tests such as false positive results, unnecessary biopsies, and
overtreatment of slow-growing cancers. His elevated PSA and subsequent
biopsy revealed a stage 1, slow-growing cancer, and he said, “one of my
options was watchful waiting, but my immediate reaction was let’s get it
out of my system.”
He considers himself lucky, however, in that the only side effect he had
from his surgery was a few months of urinary incontinence that has
since resolved.
To read the complete article : http://www.boston.com/dailydose/2012/05/21/psa-screening-for-prostate-cancer-gets-thumbs-down-from-federal-panel/ip80Gu1FRujF8B7mTGfNEJ/story.html
What do you think of the the panel's decision?
This blog is educate those interested in knowing why testing is so important in the diagnosis and treatment of cancer and other blood disorders.
Tuesday, May 22, 2012
Monday, May 21, 2012
Good Explanation of Triple Negative Breast Cancer
A wonderful explanation for us to understand 'triple negative breast cancer' is found at breastcancer.org.
To understand triple-negative breast cancer, it’s important to understand receptors, which are proteins found inside and on the surface of cells. These receptor proteins are the “eyes” and “ears” of the cells, receiving messages from substances in the bloodstream and then telling the cells what to do.
- Hormone receptors inside and on the surface of healthy breast cells receive messages from the hormones estrogen and progesterone. The hormones attach to the receptors and provide instructions that help the cells continue to grow and function well. Most, but not all, breast cancer cells also have these hormone receptors. Roughly 2 out of 3 women have breast cancer that tests positive for hormone receptors. (For a more complete explanation, see the previous section on Hormone Receptor Status.)
- A smaller percentage of breast cancers — about 20-30% — have too many HER2 receptors. In normal, healthy breast cells, HER2 receptors receive signals that stimulate their growth. With too many HER2 receptors, however, breast cancer cells grow and divide too quickly. (For a more complete explanation, see the previous section on HER2 Status.)
Hormonal therapies and HER2-targeted therapies work to interfere with the effects of hormones and HER2 on breast cancer, which can help slow or even stop the growth of breast cancer cells.
About 10-20% of breast cancers test negative for both hormone receptors and HER2 in the lab, which means they are triple-negative. Since hormones are not supporting its growth, the cancer is unlikely to respond to hormonal therapies, including tamoxifen, Arimidex (chemical name: anastrozole), Aromasin (chemical name: exemestane), Femara (chemical name: letrozole), and Faslodex (chemical name: fulvestrant). Triple-negative breast cancer also is unlikely to respond to medications that target HER2, such as Herceptin (chemical name: trastuzumab) or Tykerb (chemical name: lapatinib)
The Biology of Triple Negative Breast Cancer
In addition, triple-negative breast cancer
Tends to be more aggressive than other types of breast cancer. Studies have shown that triple-negative breast cancer is more likely to spread beyond the breast and more likely to recur (come back) after treatment. These risks appear to be greatest in the first few years after treatment. For example, a study of more than 1,600 women in Canada published in 2007 found that women with triple-negative breast cancer were at higher risk of having the cancer recur outside the breast — but only for the first 3 years. Other studies have reached similar conclusions. As years go by, the risks of the triple-negative breast cancer recurring become similar to those risk levels for other types of breast cancer.
- Tends to be higher grade than other types of breast cancer. The higher the grade, the less the cancer cells resemble normal, healthy breast cells in their appearance and growth patterns. On a scale of 1 to 3, triple-negative breast cancer often is grade 3.
- Usually is a cell type called “basal-like.” “Basal-like” means that the cells resemble the basal cells that line the breast ducts. This is a new subtype of breast cancer that researchers have identified using gene analysis technology. Like other types of breast cancer, basal-like cancers can be linked to family history, or they can happen without any apparent family link. Basal-like cancers tend to be more aggressive, higher grade cancers — just like triple-negative breast cancers. It’s believed that most triple-negative breast cancers are of the basal-like cell type.
- http://www.breastcancer.org/symptoms/diagnosis/trip_neg/behavior.jsp
Thursday, May 17, 2012
Genetic Testing: Is It Worth It?
The following video is very informational about when genetic testing should be done pertaining to cancer. This question and answer time is with a panel of oncolgists at MD Anderson Cancer Center which is one of the leading cancer centers in the United States. If you have cancer in your family then you must view this video!
Please visit: http://www.mdanderson.org/prevention for more information regarding prevention and screening
How often you should get tested for breast cancer depends on your chances for getting the disease. If you are at increased risk for breast cancer, you may need to start screening exams at an earlier age, get additional tests or be tested more often.
Learn when genetic testing makes sense for you.
Dr. Banu Arun, Professor of Breast Medical Oncology and Co-Director of Clinical Cancer Genetics at The University of Texas MD Anderson Cancer Center and Diana Turco, Certified Genetic Counselor in Clinical Cancer Genetics, answer common questions regarding hereditary risk factors for breast cancer. Dr. Jennifer Litton, Assistant Professor of Breast Medical Oncology, at The University of Texas MD Anderson Cancer Center moderates the discussion.
How often you should get tested for breast cancer depends on your chances for getting the disease. If you are at increased risk for breast cancer, you may need to start screening exams at an earlier age, get additional tests or be tested more often.
Learn when genetic testing makes sense for you.
Dr. Banu Arun, Professor of Breast Medical Oncology and Co-Director of Clinical Cancer Genetics at The University of Texas MD Anderson Cancer Center and Diana Turco, Certified Genetic Counselor in Clinical Cancer Genetics, answer common questions regarding hereditary risk factors for breast cancer. Dr. Jennifer Litton, Assistant Professor of Breast Medical Oncology, at The University of Texas MD Anderson Cancer Center moderates the discussion.
I hope that this video was very helpful and please share this with others that are concerned about their family history of cancer.
Tuesday, May 15, 2012
Primitive Neuroectodermal Tumors of the Soft Tissue or Bone
Primitive neuroectodermal tumors or PNET are very rare group of tumors that are highly malignant. In clinical manifestation these tumors exhibit great diversity. They manifest in the bone and soft tissue. Pathologist find them very difficult to distinguish because they have similarities to other small, round tumors.
PNET tumors under a microscan
PNET tumors are classified into three groups.
I. CNS primitive neuroectodermal tumors (PNETs) - Tumors derived from the central nervous system
II. Neuroblastoma - Tumors derived from the autonomic nervous system
III.Peripheral primitive neuroectodermal tumors (pPNETs) - Tumors derived from tissues outside the central and autonomic nervous system
This cancer is usually seen in children and adolescents and is very fast growing and can quickly metastasize to other organs.
Peripheral primitive neuroectodermal tumors (pPNETs) are also classified as part of the Ewing family of tumors (EFTs); peripheral primitive neuroectodermal tumors (pPNETs) and Ewing family of tumors (EFTs) are often referred to interchangeably in the literature. Generally, Ewing family of tumors (EFTs) and peripheral primitive neuroectodermal tumors (pPNETs) represent different manifestations of the same tumor and have similar genetic alterations. Ewing sarcoma, however, is more common in bone, while peripheral primitive neuroectodermal tumors (pPNETs) are more common in soft tissues. Immunohistochemical and cytogenetic studies suggest that these tumors all have a common origin.
Tissue biopsy with cytogenetic and immunohistochemical studies is paramount in diagnosing peripheral primitive neuroectodermal tumors (pPNETs). Radiologic studies such as CT scans and MRI are essential in determining the limits of tumor involvement and ruling out metastatic disease. On CT scans, peripheral primitive neuroectodermal tumors (pPNETs) appear as heterogeneous masses, often invading surrounding tissues, including bone. MRI reveals a mass isointense to muscle on T1-weighted images, while hyperintense on T2-weighted image
Current recommendations advocate complete surgical resection whenever possible, adjuvant versus neoadjuvant chemotherapy, and radiotherapy. Multimodality treatment is advocated to prevent metastatic disease, recurrent disease, and to treat residual tumor after resection. Carvajal and Meyers, in a comprehensive review of the chemotherapeutic regimens in the treatment of PNETs and Ewing family of
tumors (EFTs), recommend a regimen that includes vincristine, doxorubicin, and cyclophosphamide with ifosfamide and etoposide.
For more specific information:
http://emedicine.medscape.com/article/855644-overview#aw2aab6b8
PNET tumors under a microscan
PNET tumors are classified into three groups.
I. CNS primitive neuroectodermal tumors (PNETs) - Tumors derived from the central nervous system
II. Neuroblastoma - Tumors derived from the autonomic nervous system
III.Peripheral primitive neuroectodermal tumors (pPNETs) - Tumors derived from tissues outside the central and autonomic nervous system
This cancer is usually seen in children and adolescents and is very fast growing and can quickly metastasize to other organs.
Peripheral primitive neuroectodermal tumors (pPNETs) are also classified as part of the Ewing family of tumors (EFTs); peripheral primitive neuroectodermal tumors (pPNETs) and Ewing family of tumors (EFTs) are often referred to interchangeably in the literature. Generally, Ewing family of tumors (EFTs) and peripheral primitive neuroectodermal tumors (pPNETs) represent different manifestations of the same tumor and have similar genetic alterations. Ewing sarcoma, however, is more common in bone, while peripheral primitive neuroectodermal tumors (pPNETs) are more common in soft tissues. Immunohistochemical and cytogenetic studies suggest that these tumors all have a common origin.
Tissue biopsy with cytogenetic and immunohistochemical studies is paramount in diagnosing peripheral primitive neuroectodermal tumors (pPNETs). Radiologic studies such as CT scans and MRI are essential in determining the limits of tumor involvement and ruling out metastatic disease. On CT scans, peripheral primitive neuroectodermal tumors (pPNETs) appear as heterogeneous masses, often invading surrounding tissues, including bone. MRI reveals a mass isointense to muscle on T1-weighted images, while hyperintense on T2-weighted image
Due to the fact that PNET is metastatic then full body scans need to be performed to see the extent of the tumor growth.
Treatment Plan
Chemotherapy and radiation are necessary adjuncts in the treatment of primitive neuroectodermal tumors (PNETs). Chemotherapy regimens have significantly improved outcomes in patients with peripheral primitive neuroectodermal tumors (pPNETs). The treatment paradigms differ based on whether the disease is localized or metastatic. As would be expected, the treatments for peripheral primitive neuroectodermal tumors (pPNETs) and Ewing family of tumors (EFTs) are similar in terms of chemotherapeutic regimens.Current recommendations advocate complete surgical resection whenever possible, adjuvant versus neoadjuvant chemotherapy, and radiotherapy. Multimodality treatment is advocated to prevent metastatic disease, recurrent disease, and to treat residual tumor after resection. Carvajal and Meyers, in a comprehensive review of the chemotherapeutic regimens in the treatment of PNETs and Ewing family of
tumors (EFTs), recommend a regimen that includes vincristine, doxorubicin, and cyclophosphamide with ifosfamide and etoposide.
For more specific information:
http://emedicine.medscape.com/article/855644-overview#aw2aab6b8
Monday, May 14, 2012
Not All Women are Treated the Same With Ovarian Cancer
Researcher finds problems with the fact that all women with ovarian cancer do not have access to top quality care to boost survival.
Dr. Robert Bristow believes a decidedly low-tech approach could significantly enhance the survival rate for ovarian cancer, even though it’s the deadliest women’s reproductive cancer, claiming 15,000 lives each year; it has no reliable screening or prevention methods; and its research funding is about one-sixth the amount for breast cancer.
According to CancerCenter.com recent study and interview there only need to be excellent care for all women concerning ovarian cancer.
“We don’t have to redesign a molecule to improve the outcome for women with ovarian cancer,” says Bristow, the Philip J. DiSaia Chair in Gynecologic Oncology and director of UC Irvine’s Division of Gynecologic Oncology. “Recent research has shown that the most profound impact on survivorship occurs when women get proper care from surgeons trained in the latest techniques for treating ovarian cancer.”
The sad news is that it was discovered that women with low income and black women did not receive the excellent care as white women or affluent women. This study was done in March of 2012. Our health plans have got to change.
“Not all women are benefiting equally from improvements in ovarian cancer care,” Bristow says. “The reasons behind these disparities are not entirely clear, which is why we need additional research.”
For the women who did not receive excellent care due to race and social standing they also extended the research to see if when they were treated for ovarian cancer if the they followed the National Comprehensive Cancer Network treatment guidelines.
Bristow and colleagues found that five-year survival rates varied significantly. (Improvement in ovarian cancer care is measured in length of survival after diagnosis rather than a “cure” rate.)
Among those whose care met NCCN standards, the rate for white women was 41.4 percent, compared with 33.3 percent for African American women. Among those whose care did not meet NCCN standards, the rate for white women was 37.8 percent, compared with 22.5 percent for African American women. Those on Medicaid or without insurance faced a 30 percent increased risk of death. Poor women – defined as having an annual household income of less than $35,000 – had worse survival rates regardless of race.
http://medicalxpress.com/news/2012-05-ovarian-cancer.html
Dr. Robert Bristow believes a decidedly low-tech approach could significantly enhance the survival rate for ovarian cancer, even though it’s the deadliest women’s reproductive cancer, claiming 15,000 lives each year; it has no reliable screening or prevention methods; and its research funding is about one-sixth the amount for breast cancer.
According to CancerCenter.com recent study and interview there only need to be excellent care for all women concerning ovarian cancer.
“We don’t have to redesign a molecule to improve the outcome for women with ovarian cancer,” says Bristow, the Philip J. DiSaia Chair in Gynecologic Oncology and director of UC Irvine’s Division of Gynecologic Oncology. “Recent research has shown that the most profound impact on survivorship occurs when women get proper care from surgeons trained in the latest techniques for treating ovarian cancer.”
The sad news is that it was discovered that women with low income and black women did not receive the excellent care as white women or affluent women. This study was done in March of 2012. Our health plans have got to change.
“Not all women are benefiting equally from improvements in ovarian cancer care,” Bristow says. “The reasons behind these disparities are not entirely clear, which is why we need additional research.”
For the women who did not receive excellent care due to race and social standing they also extended the research to see if when they were treated for ovarian cancer if the they followed the National Comprehensive Cancer Network treatment guidelines.
Bristow and colleagues found that five-year survival rates varied significantly. (Improvement in ovarian cancer care is measured in length of survival after diagnosis rather than a “cure” rate.)
Among those whose care met NCCN standards, the rate for white women was 41.4 percent, compared with 33.3 percent for African American women. Among those whose care did not meet NCCN standards, the rate for white women was 37.8 percent, compared with 22.5 percent for African American women. Those on Medicaid or without insurance faced a 30 percent increased risk of death. Poor women – defined as having an annual household income of less than $35,000 – had worse survival rates regardless of race.
http://medicalxpress.com/news/2012-05-ovarian-cancer.html
Saturday, May 12, 2012
Sharing Article of Breast Cancer Diagnostic Tool
This is great news that was published this past week and it must be shared with all women in the fight against breast cancer. Please share this article with a friend .
Below is an image of detecting chromosome translocations in sections of breast.
In breast cancer, we have shown that the NRG1/heregulin gene is translocated in 6% of primary cases and Soda and colleagues described fusions of ALK in 7% of lung cancers .
“HER-2/neu FISH testing is considered the gold standard because it has the advantage of looking at each tumor cell individually,” said Olga Falkowski, MD, a board-certified pathologist who serves as the Associate Medical Director and Director of Genetics at Acupath Laboratories, Inc., a national medical laboratory based in Plainview, NY. “Under U.S. Food & Drug Administration guidelines, all women with invasive breast cancer are eligible to undergo the test.”
In the body, the HER protein is a normally occurring substance that helps cells grow and stay strong. In about 20 percent of breast cancers, patients are HER-2 positive, meaning that cancer cells underwent genetic mutations responsible for production of excess protein called the HER-2/neu or human epidermal growth factor receptor 2. Patients with the genetic mutation HER-2 in breast cancer are eligible for a targeted therapy. Because of this, both the American Society for Clinical Oncology and National Comprehensive Cancer Network recommend HER-2/neu testing for all breast cancer tumors.
Women who undergo a biopsy for a suspicious lump and are diagnosed with cancer first receive immunohistochemistry or IHC of HER-2/neu testing as part of their initial laboratory workup. If the IHC testing is positive for Her-2/neu, then FISH is performed to confirm the genetic mutation.
What makes FISH or fluorescence in situ hybridization testing different and generally more effective, Dr. Falkowski said, is that it examines each individual cell of the tumor. “That’s a huge advantage,” she added. “The more a doctor knows about a tumor and its characteristics, the better that doctor can determine the most effective treatment options. The HER-2/neu FISH test tends to be more reliable.”
IHC testing quality can vary between laboratories based on the type of antibodies the lab uses, the way the lab prepares the tissue sample, and its criteria for determining the presence of HER-2, which is not the same everywhere. “Different labs have different reading systems,” Dr. Falkowski explained.
FISH testing is a more complex procedure, but is more specific and sensitive. It uses fluorescent probes to identify, tag and count the presence of genes that cause excessive production of the HER-2 protein in each individual cell. If more than two copies of the gene are found in each cell, the cancer is determined to be HER-2 positive and treated as such.
“It’s a more reproducible test, meaning the results from one lab to another should be the same,” Dr. Falkowski explained, adding that the HER-2 protein can also affect the aggressiveness and treatment of gastric and ovarian cancer. “Because it’s expensive technology, HER-2/neu FISH testing isn’t available everywhere, so doctors and their patients should know to ask for it,” adds Dr. Falkowski.
A concrete, reliable diagnosis of whether a woman with breast cancer is HER-2 positive will help lead to the best possible treatment starting immediately, improving survival and helping prevent recurrence. Standard chemotherapy drugs can be effective in treating Her-2/neu-positive patients, though more effective are two drugs that specifically target the Her-2/neu protein, trastuzumab (Herceptin) and lapatinib (Tykerb). Trastuzumab can be used alone or in conjunction with chemotherapy or hormone-blocking medications, though it comes with some potentially risky side effects, including congestive heart failure. Lapatinib is also generally used in combination with chemotherapy, most often in patients who have problems with trastuzumab. A promising new drug called trastuzumab emtansine or T-DM1 that combines several agents is also currently in clinical trials.
Dr. Falkowski continued, “As dramatic as this may sound, it’s true: For some women, HER-2/neu FISH testing could mean the difference between life and death.”
Olga Falkowski, M.D. is board-certified in anatomic and clinical pathology by the American Board of Pathology, and serves as the Associate Medical Director and Director of Genetics at Acupath Laboratories, Inc.
Acupath Laboratories, Inc. is a Plainview, New York, specialty medical lab engaged in cutting-edge diagnostics. http://www.acupath.com
http://www.prweb.com/releases/2012/5/prweb9457999.htm
Below is an image of detecting chromosome translocations in sections of breast.
In breast cancer, we have shown that the NRG1/heregulin gene is translocated in 6% of primary cases and Soda and colleagues described fusions of ALK in 7% of lung cancers .
Article
Pathologists today know that there are certain gene mutations that are predictive of cancers; for example, women with the BRCA1 or BRCA2 mutation are at a greater risk of developing breast and/or ovarian cancer. What is less known is the availability of genetic HER-2/neu FISH testing, a sophisticated laboratory test able to better determine treatment options for an aggressive form of breast cancer. This information is important because it helps doctors to determine the best course of treatment for each patient.“HER-2/neu FISH testing is considered the gold standard because it has the advantage of looking at each tumor cell individually,” said Olga Falkowski, MD, a board-certified pathologist who serves as the Associate Medical Director and Director of Genetics at Acupath Laboratories, Inc., a national medical laboratory based in Plainview, NY. “Under U.S. Food & Drug Administration guidelines, all women with invasive breast cancer are eligible to undergo the test.”
In the body, the HER protein is a normally occurring substance that helps cells grow and stay strong. In about 20 percent of breast cancers, patients are HER-2 positive, meaning that cancer cells underwent genetic mutations responsible for production of excess protein called the HER-2/neu or human epidermal growth factor receptor 2. Patients with the genetic mutation HER-2 in breast cancer are eligible for a targeted therapy. Because of this, both the American Society for Clinical Oncology and National Comprehensive Cancer Network recommend HER-2/neu testing for all breast cancer tumors.
Women who undergo a biopsy for a suspicious lump and are diagnosed with cancer first receive immunohistochemistry or IHC of HER-2/neu testing as part of their initial laboratory workup. If the IHC testing is positive for Her-2/neu, then FISH is performed to confirm the genetic mutation.
What makes FISH or fluorescence in situ hybridization testing different and generally more effective, Dr. Falkowski said, is that it examines each individual cell of the tumor. “That’s a huge advantage,” she added. “The more a doctor knows about a tumor and its characteristics, the better that doctor can determine the most effective treatment options. The HER-2/neu FISH test tends to be more reliable.”
IHC testing quality can vary between laboratories based on the type of antibodies the lab uses, the way the lab prepares the tissue sample, and its criteria for determining the presence of HER-2, which is not the same everywhere. “Different labs have different reading systems,” Dr. Falkowski explained.
FISH testing is a more complex procedure, but is more specific and sensitive. It uses fluorescent probes to identify, tag and count the presence of genes that cause excessive production of the HER-2 protein in each individual cell. If more than two copies of the gene are found in each cell, the cancer is determined to be HER-2 positive and treated as such.
“It’s a more reproducible test, meaning the results from one lab to another should be the same,” Dr. Falkowski explained, adding that the HER-2 protein can also affect the aggressiveness and treatment of gastric and ovarian cancer. “Because it’s expensive technology, HER-2/neu FISH testing isn’t available everywhere, so doctors and their patients should know to ask for it,” adds Dr. Falkowski.
A concrete, reliable diagnosis of whether a woman with breast cancer is HER-2 positive will help lead to the best possible treatment starting immediately, improving survival and helping prevent recurrence. Standard chemotherapy drugs can be effective in treating Her-2/neu-positive patients, though more effective are two drugs that specifically target the Her-2/neu protein, trastuzumab (Herceptin) and lapatinib (Tykerb). Trastuzumab can be used alone or in conjunction with chemotherapy or hormone-blocking medications, though it comes with some potentially risky side effects, including congestive heart failure. Lapatinib is also generally used in combination with chemotherapy, most often in patients who have problems with trastuzumab. A promising new drug called trastuzumab emtansine or T-DM1 that combines several agents is also currently in clinical trials.
Dr. Falkowski continued, “As dramatic as this may sound, it’s true: For some women, HER-2/neu FISH testing could mean the difference between life and death.”
Olga Falkowski, M.D. is board-certified in anatomic and clinical pathology by the American Board of Pathology, and serves as the Associate Medical Director and Director of Genetics at Acupath Laboratories, Inc.
Acupath Laboratories, Inc. is a Plainview, New York, specialty medical lab engaged in cutting-edge diagnostics. http://www.acupath.com
http://www.prweb.com/releases/2012/5/prweb9457999.htm
Friday, May 11, 2012
Metastatic Breast Cancer
Metastatic breast cancer is a stage of breast cancer where the disease has spread to distant metastases.
It is a complication of primary breast cancer, usually occurring
several years after resection of the primary breast cancer. Metastatic
breast cancer cells frequently differ from the preceding primary breast
cancer in properties such as receptor status, have often developed
resistance to several lines of previous treatment and acquired special
properties that permit them metastasize to distant sites, making them
especially dangerous. The prognosis is often poor, distant metastases
are the cause of about 90% of deaths due to breast cancer.
Breast cancer primarily metastasizes to the bone, lungs, regional lymph nodes, liver and brain, with the most common site being the bone. Lymph node metastsasis into the sentinel node and few surrounding nodes is regarded as a treatable local event and not metastatic breast cancer, both when occurring at primary presentation or later.
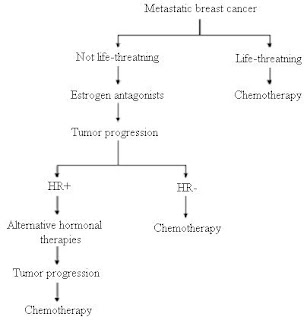
Progression of Metastatic Breast Cancer
Advancements in treating metastatic cancer by Professor Steven Twelves M.D.
Faces of those with Metastatic Cancer
Today, say the MD Anderson researchers, as many as 40% of women with recurrent or metastatic breast cancer survive at least five years. "More and more, both doctors and patients approach it as a chronic condition," says Eric Winer, MD, director of the Breast Program at Boston's Dana-Farber Cancer Institute. "We can't cure it, but we can manage it for many years."
For more information:http://www.webmd.com/breast-cancer/features/metastatic-breast-cancer-chronic-condition
Breast cancer primarily metastasizes to the bone, lungs, regional lymph nodes, liver and brain, with the most common site being the bone. Lymph node metastsasis into the sentinel node and few surrounding nodes is regarded as a treatable local event and not metastatic breast cancer, both when occurring at primary presentation or later.
Progression of Metastatic Breast Cancer
Today, say the MD Anderson researchers, as many as 40% of women with recurrent or metastatic breast cancer survive at least five years. "More and more, both doctors and patients approach it as a chronic condition," says Eric Winer, MD, director of the Breast Program at Boston's Dana-Farber Cancer Institute. "We can't cure it, but we can manage it for many years."
For more information:http://www.webmd.com/breast-cancer/features/metastatic-breast-cancer-chronic-condition
Subscribe to:
Posts (Atom)